
McGill Univ. Libraries and the generosity of Dr. Richard L. Tomlinson
 |
Brought to you by: McGill Univ. Libraries and the generosity of Dr. Richard L. Tomlinson |
|||
|
|
|
| Bioorganic
& Medicinal Chemistry Letters Volume 15, Issue 4, 15 February 2005, Pages 1135-1138 |
| ||||
|
|
| ||||
Synthesis of half-mustard combi-molecules with fluorescence properties: correlation with EGFR status
Zakaria Rachid, Fouad Brahimi, Juozas Domarkas and Bertrand Jacques
Jean-Claude![]() ,
, ![]()
Abstract
The synthesis of 6-(2-chloroethylamino)-4-anilinoquinazolines ZR2002 and
ZR2003 designed to block EGFR tyrosine kinase and to damage genomic DNA is
described. These compounds present fluorescence properties that permitted the
quantitation of their subcellular uptake by flow cytometry. Fluorescence
intensities increased with increasing levels of EGFR in a panel of isogenic and
established cell lines.
Graphical abstract
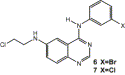
The synthesis of 6-(2-chloroethylamino)-4-anilinoquinazolines (ZR2002 and
ZR2003) designed to block EGFR tyrosine kinase and to damage genomic DNA is
described. These compounds present fluorescence properties that permitted the
quantitation of their subcellular uptake by flow cytometry. Fluorescence
intensities increased with increasing levels of EGFR in a panel of isogenic and
established cell lines.
 |
Display Full Size version of this image (6K) |
Overexpression of the epidermal growth factor receptor (EGFR) and its closest homologue HER2 is associated with aggressive tumour progression and reduced sensitivity to antitumour agents.1, 2, 3 and 4 Blockade of the tyrosine kinase activity of EGFR and the HER2 gene product p185neu induces significant antitumour activity in vivo and Iressa™, an aminoquinazoline that binds to the ATP site of EGFR has proven efficacious against many human tumours in nude mice.5 This is now approved for the treatment of lung cancer.6 Thus, the detection of EGFR and HER2 in tumour biopsies is now investigated as a useful strategy for both diagnosis and treatment. Since in many cases, inhibition of EGFR does not suffice to eradicate the tumour, we recently developed a novel class of molecules termed combi-molecules or TZ-I capable of not only blocking EGFR-mediated signalling by binding to its ATP site, but also damaging DNA.
Fortuitously, this class of compounds showed fluorescence property that allowed us to explore their development as non-immunological probes for the detection of EGFR. Due to the high cost of monoclonal antibodies, the rapid small-molecule staining of EGFR/p185neu would present a major clinical advantage.
The development of the molecules described herein stems from our novel ‘combi-targeting’ strategy that seeks to design and study agents termed ‘combi-molecules’ or TZ-I (see Scheme 1) ‘programmed’ to degrade to an inhibitor of EGFR (I) and a DNA damaging species (TZ)7, 8, 9, 10 and 11 (see path 1). However, the study of combi-molecules or TZ-I capable of blocking EGFR phosphorylation and damaging DNA without hydrolytic cleavage remained to be achieved (see path 2). More recently, we demonstrated that ZR2002 (6) was the first prototype of combi-molecule capable of generating the binary EGFR/DNA targeting activity without requirement for hydrolytic cleavage.12 Here we describe the synthesis of this compound and correlated its total cellular uptake with the EGFR status of a panel of human tumour cells.
Display Full Size version of this image (7K) Scheme 1.
The synthesis of 6 and its 3′-chloro analogue 7 proceeded as described in Scheme 2. Briefly, compound 2 was obtained as described by Roth and Tai13 by treating the 5-nitroanthranilonitrile 1 with a sulfuric acid/formic acid mixture. The resulting quinazoline 2 was heated with phosphorous pentachloride to provide 3, which was treated with substituted anilines. The nitro group of the resulting compounds was reduced with Fe in ethanol as previously described14, 15, 16, 17 and 18 to give the amines 4 and 5. Diazotization of aminoquinazolines 4–5 using NOBF4 in acetonitrile, followed by the addition of chloroethylamine gave 6 and 719 as pure solids after neutralization with triethylamine and purification by column chromatography on basic alumina. Their structures were confirmed by 1H, 13C NMR and high resolution mass spectrometry (HRMS). In order to elucidate the mechanism of formation of 6–7, the reaction was carried out in CD3CN and neutralization performed with K2CO3 in D2O. 1H NMR analysis (see Fig. 1) showed that the transient triazene species is predominantly the non-conjugated tautomer 9. Interestingly, intramolecular cyclization may have led to the formation of the 1,2,3-triazole 10. The structures were assigned on the basis of NMR data for similar compounds.20 Thus, the proposed mechanism for the formation of 6–7 is depicted in Scheme 3. Under basic conditions the predominant tautomeric form 9 collapsed to 6–7 following loss of nitrogen (most likely through a radical mechanism) or perhaps cyclized to give 10. It should be noted that when the mixtures obtained from the coupling reactions with 2-chloroethylamine were loaded on the alumina column, we were only able to isolate compounds 6 and 7 (Scheme 2).
Display Full Size version of this image (25K) Scheme 2. (i) H2SO4/formic acid/heat; (ii) PCl5/heat; (iii) ArNH2/iPrOH/rt; (iv) Fe/ethanol/AcOH/heat; (v) CH3CN/NOBF4/−5 °C/ether then addition of Et3N/2-chloroethylamine hydrochloride at rt overnight.
Display Full Size version of this image (14K) Figure 1. NMR spectrum of the reaction mixture after treatment of 5 with NOBF4, addition of 2-chloroethylamine hydrochloride in CD3CN and neutralization with K2CO3/D2O.
Display Full Size version of this image (22K) Scheme 3.
On the other hand, in addition to their significant EGFR binding affinity (Table 1), structures 6–7 were found to possess significant fluorescence properties. Indeed at maximum absorption (270 nm), fluorescence emission at 451 nm was observed (Fig. 2). Since recent studies with ZR2002 demonstrated that it is an irreversible inhibitor of EGFR11 we surmised that it could be used to probe EGFR levels in the cells. More importantly, its ability to covalently bind to intracellular macromolecules would promote intracellular retention. It should be remembered herein that the irreversible mechanism of action of compounds bearing reactive group at the 6-position of the quinazoline ring was attributed to their ability to alkylate Cys-773 of EGFR.21 and 22 Thus, to test the ability of these molecules to probe the EGFR content of tumour cells, we analyzed fluorescence emission following cell exposure to low concentration of 6. Fluorescence intensities/single cell were measured by flow cytometry23 and correlated with EGFR levels quantitated by western blotting using anti-EGFR antibody staining. The analysis was performed in a panel of cells harbouring varied levels of EGFR and isogenic pairs of lines stably transfected with EGFR. A statistically significant linear correlation (Pearson correlation coefficient: r = 0.7, p < 0.02) was observed. Although the correlation was statistically significant, a better linearity of the curve would be required for a potential application of this observation to diagnostics. The lack of linearity of the correlation may be due to unspecific binding of the inhibitor to other structural homologue of EGFR or related proteins. Nevertheless, this work sets premise for the development of more specific EGFR inhibitor with fluorescence property for the development of inexpensive non biologic detection method for EGFR in the clinic.
Inhibition of EGFR tyrosine kinase by different quinazolines
a IC50 to inhibit the phosphorylation of poly(L-glutamic acid-L-tyrosine) by EGFR isolated from A431 carcinoma cells.
Compounds EGFR TK Inhibition IC50, μMa 4 0.0447 5 0.217 6 0.0112 7 0.026
Display Full Size version of this image (15K) Figure 2. Correlation between the levels of EGFR and single-cell fluorescence intensity in a panel of human tumour cell lines as measured by flow cytometry.
Acknowledgements
We thank the National Cancer Institute of Canada (NCIC) (Grant #: 01377) for financial support.
References and notes
1 H. Meden and W. Kuhn, Eur. J. Obste. Gyne Reprod. Biol. 71 (1997), p. 173. SummaryPlus | Full Text + Links | PDF (641 K) | View Record in Scopus | Cited By in Scopus (64)
2 H. Modjtahedi, Dean. Int. J. Oncol. 4 (1998), p. 277.
3 C. Xinmei, T.K. Yeung and Z. Wang, Biochem. Bioph. Res. Com. 277 (2000), p. 757.
4 S.S. Hsieh, C. Malerczyk, A. Aigner and F. Czubayko, J. Cancer 86 (2000), p. 644. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (25)
5 F. Giardiello, F. Caputo, R. Bianco, V. Damianco, G. Pomatico, S. De placido, A.R. Bianco and G. Tortora, Clin. Cancer Res. 6 (2001), p. 2053.
6 W. Pao, V.A. Miller, E. Venkatraman and M.G. Kris, J. Nat. Cancer. Ins. 96 (2004), p. 1117. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (17)
7 S.L. Matheson, J.P. McNamee and B.J. Jean-Claude, Biochem Pharmacol. 67 (2004), p. 1131. SummaryPlus | Full Text + Links | PDF (385 K) | View Record in Scopus | Cited By in Scopus (8)
8 S.L. Matheson, J. McNamee and B.J. Jean-Claude, J. Pharmacol. Exp. Ther. 296 (2001), p. 832. View Record in Scopus | Cited By in Scopus (29)
9 F. Brahimi, S.L. Matheson, F. Dudouit, J.P. McNamee, A.M. Tari and B.J. Jean-Claude, J. Pharmacol. Exp. Ther. 303 (2002), pp. 238–246. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (22)
10 S.L. Matheson, J. McNamee and B.J. Jean-Claude, Cancer Chemother. Pharmacol. 51 (2003), pp. 11–20. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (19)
11 Q. Qiu, F. Dudouit, S.L. Matheson, F. Brahimi, R. Banerjee, J.P. McNamee and B.J. Jean-Claude, Cancer Chemother. Pharmacol. 51 (2003), pp. 1–10. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (19)
12 F. Brahimi, Z. Rachid, J.P. McNamee, Y. Li, A.M. Tari and B.J. Jean-Claude, Int. J. Cancer 112 (2004), pp. 484–491. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (6)
13 G.A. Roth and J.J. Tai, J. Heterocyclic Chem. 33 (1996), pp. 2051–2053. View Record in Scopus | Cited By in Scopus (7)
14 G.W. Rewcastle, D.K. Murray, W.L. Elliott, D.W. Fry, C.T. Howard, J.M. Nelson, B.J. Roberts, P.W. Vincent, H.D. Showalter, T.R. Winters and W.A. Denny, J. Med. Chem. 41 (1998), pp. 742–751. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (75)
15 G.W. Rewcastle, W.A. Denny, A.J. Bridges, Z. Hairong, D.R. Cody, A. McMichael and D.W. Fry, J. Med. Chem. 38 (1995), pp. 3482–3487. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (125)
16 G.W. Rewcastle, A.J. Bridges, D.W. Fry, J.R. Rubin and W.A. Denny, J. Med. Chem. 40 (1997), pp. 1820–1826. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (46)
17 Z. Rachid, F. Brahimi, A. Katsoulas, N. Teo and B.J. Jean-Claude, J. Med. Chem. 46 (2003), pp. 4313–4321. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (12)
18 B. Banerejee, Z. Rachid, J. McNamee and B.J. Jean-Claude, J. Med. Chem. 46 (2003), pp. 5546–5551.
N-(2-Chloroethyl)-N-(3-bromophenyl)-quinazoline-4,6-diamine (6). The amino compound 4 (1.26 g, 0.4 mmol) was stirred in dry acetonitrile (40 mL) under argon, after which the solution was cooled to −5 °C followed by the addition of nitrosonium tetrafluoroborate (0.90 g, 0.8 mmol) in acetonitrile. The resulting clear solution was stirred for 1 h at −5 °C to permit the formation of the diazonium salt. It was then added dropwise to another solution of ether (30 mL), water (5 mL), Et3N (6 mL) and 2-chloroethylamine hydrochloride (3.24 g, 2.8 mmol) at 0 °C. The mixture was stirred at room temperature overnight followed by the subsequent extraction with ethyl acetate. The organic layer was dried over potassium carbonate and evaporated to provide the crude product, which was purified by chromatography using a basic alumina column (1:4 triethylamine–EtOAc) to give an oil that solidified upon addition of petroleum ether and ether to give 6 (400 mg, 27%); mp 167 °C; ESI m/z 377.3 (MH+ with 79Br), 379.2 (MH+ with 81Br), 381.2 (MH+ with 81Br, 37Cl), 341.3 (M–Cl, 16.68) m/z 376.00973 (M), C16H14BrClN4 requires m/z 376.00904; 1H NMR (400 MHz, DMSO-d6) δ 9.48 (s, 1H, NH), 8.39 (s, 1H, H-2), 8.12 (s, 1H, H-2′), 7.86 (dd, 1H, J = 1.2 Hz, J = 10.8 Hz, H-6′), 7.55 (1H, d, J = 12.4 Hz, H-8), 7.36–7.25 (m, 4H, H-5-7-4′-5′), 6.52 (t, 1H, J = 8 Hz, NH), 3.85 (t, 2H, J = 7.6 Hz, CH2–Cl), 3.62 (q, 2H, J = 8 Hz, CH2); 13C NMR (100 MHz, DMSO-d6) d 156.4, 150.3, 147.5, 143.0, 141.9, 131.0, 129.0, 126.4, 124.9, 124.8, 121.8, 121.5, 117.1, 97.6, 45.6, 43.9
20 Z.B. Papanastassiou, R.J. Bruni, E.V. White and P.L. Levins, J. Med. Chem. 9 (1966), pp. 725–729. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (3)
21 D.W. Fry, A.J. Bridges, W.A. Denny, A. Doherty, K.D. Greis, J.L. Hicks, K.E. Hook, P.R. Keller, W.R. Leopold, J.A. Loo, D.J. McNamara, J.M. Nelson, V. Sherwood, J.B. Smaill, S. Trumpp-Kallmeyer and E.M. Dobrusin, Proc. Natl. Acad. Sci. U.S.A. 95 (1998), pp. 12022–12027. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (187)
22 L.F. Allen, P.F. Lenehan, I.A. Eiseman, W.L. Elliott and D.W. Fry, Sem. Oncol. 29 (2002), pp. 11–21. View Record in Scopus | Cited By in Scopus (73)
Flow cytometric analysis: Cells were grown in 6-well plates until confluence
and then incubated with ZR2002 for 30 min. Thereafter, they were harvested
by trypsin-EDTA, collected by centrifugation and washed twice with PBS. Analyses
were performed by flow cytometry using UV filter of 424 ± 44 nm.
The following cell lines were analyzed: SF-126, SF-126-EGFR, SF-188, SF188-EGFR,
MDA-MB-468, MDA-MB-231, MDA-MB-435, A431
| Bioorganic
& Medicinal Chemistry Letters Volume 15, Issue 4, 15 February 2005, Pages 1135-1138 |
|
|
 |